Zimmer Knee Replacements
The Zimmer Knee is a popular knee replacement device for people with painful, damaged knees. Some people who received a Zimmer Knee have experienced premature loosening of the device, leading to intense pain and revision surgery.
What are Zimmer Knee Replacements?
The Zimmer Knee is an artificial knee made of plastic and metal components. It replaces damaged or degenerated knee cartilage caused by osteoarthritis, rheumatoid arthritis and knee trauma. Some people who had a knee replacement with these products had to have a follow-up surgery when the replacement knee loosened prematurely, which has led to Zimmer Knee lawsuits
In 1995, the manufacturer of the Zimmer Knee introduced its flagship project, the NexGen Complete Knee Solution. Building off the success of the NexGen replacement knees, the company released its line of “high-flex” NexGen replacement knees in 2001. The high-flex NexGen knee replacements were designed to offer a greater range of motion than standard knee replacements.
The Zimmer NexGen Knee is the most widely sold line of artificial knee replacements in the world, accounting for 25 percent of all knee replacements.
Zimmer Biomet, the maker of the NexGen Knee, manufactures thousands of products sold across the world. The company’s gross profit in 2013 was approximately $2.4 billion. In 2010, sales from the Zimmer Knee accounted for 42 percent of the company’s revenue.
Some evidence suggests the high-flex NexGen Knees may loosen earlier than standard knee replacements. Specific products in the NexGen line people have struggled with include:
- NexGen Complete Knee Solution Legacy Posterior Stabilized-Flex Femoral Components (LPS-Flex)
- NexGen Complete Knee Solution Cruciate Retaining-Flex Femoral Components (CR-Flex)
- NexGen Complete Knee Solution Gender Solutions Female LPS-Flex (GSF LPS-Flex)
- NexGen Complete Knee Solution CR-Flex Gender Solutions Female CR-Flex (GSF-Flex)
- All NexGen MIS (Minimally Invasive Surgery) Total Knee Procedure Stemmed Tibial Components
How do Zimmer Knee Replacements work?
The knee is the point where the femur (thigh bone) and the tibia (shin bone) meet. These bones are held together by muscles, ligaments and tendons. Cartilage layers the ends of the bone, acting as a shock absorber. Pain and immobility results when the cartilage breaks down.
Knee replacement surgery, also known as arthroplasty, helps restore mobility and relieve joint pain. An arthroplasty relieves pain and stiffness in the knee by replacing the damaged or degenerated parts of the joint. It is the last option when other treatments, such as therapy and medication, are no longer effective.
In a knee replacement surgery, doctors make an incision (8 to 10 inches) in front of the knee. A surgeon then removes the damaged parts of the knee joint from the surface of the bone. The surgeon shapes and smoothes the surface of the bone to mold with the artificial knee.
Doctors use metal components to cap the ends of the femur and tibia. These caps are usually attached with surgical cement. A plastic component replaces the damaged cartilage. The new knee joint creates a pain-free surface that glides easier.
Zimmer Knee Replacement Side Effects
 Zimmer designed NexGen Knee to allow better knee flexibility than other artificial knees. However, some studies suggest that the NexGen Knee isn’t much better than a standard knee replacement, which may have fewer complications.
Zimmer designed NexGen Knee to allow better knee flexibility than other artificial knees. However, some studies suggest that the NexGen Knee isn’t much better than a standard knee replacement, which may have fewer complications.
A study published in the Bone & Joint Journal stated that the NexGen LPS-Flex “showed a marked rate of early loosening of the femoral component.”
Artificial knees should last anywhere from 10 to 20 years. Yet, some people with a Zimmer NexGen Flex Knee have experienced premature failure and loosening of the the device in as little as one or two years. If the device fails, the patient is usually readmitted for surgery.
The loosening of a knee implant can lead to:
Device Complications
- Premature failure
- Loosening of the device
- Stiffness
Health Consequences
- Pain
- Swelling
- Limiting range of motion
- Limping
- Instability
- Bone loss around failed knee replacement
Evidence of Zimmer Knee Replacement Complications
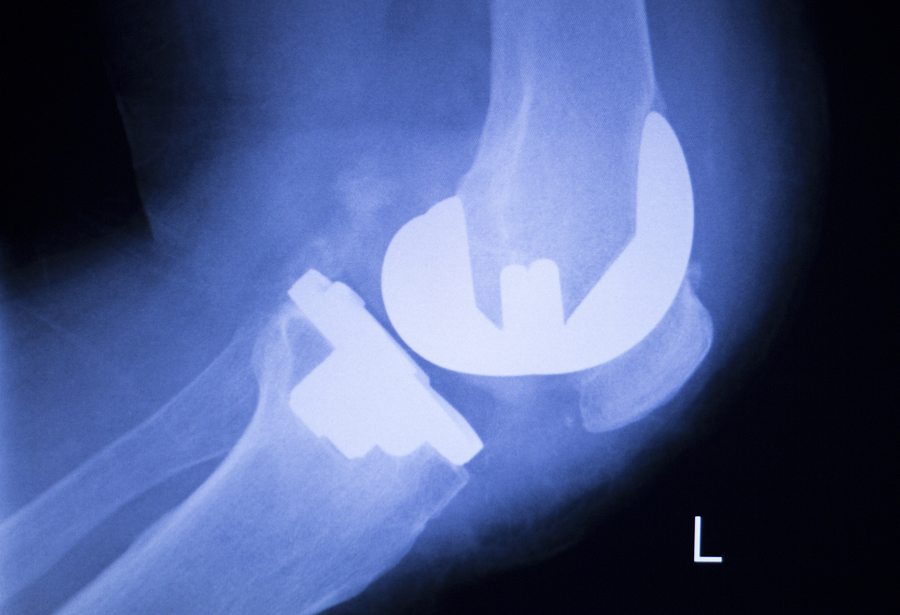 The FDA approved the first NexGen Knees in 1999 through a 510(k) premarket notification process. This premarket process allows companies to release medical devices without testing them if the device is similar to devices already on the market.
The FDA approved the first NexGen Knees in 1999 through a 510(k) premarket notification process. This premarket process allows companies to release medical devices without testing them if the device is similar to devices already on the market.
Zimmer claimed the design of their NexGen knee was identical to their earlier artificial knees except for small changes to improve the NexGen’s range of motion.
The authors of a 2015 study published in the Journal of Arthroplasty stated doubts about NexGen’s increased flexion. They said that their findings “do not support the proposition that high flexion knee prostheses provide substantial clinical advantages over standard knee prostheses.” Another study found that half of the participants experienced loosening of their knee replacement.
Resources for Zimmer Knee Replacement Complications
If you or a loved one have experienced the negative consequences of a Zimmer Knee replacement premature failure, you may be entitled to compensation for lost wages and medical bills.
Last Edited: November 1, 2017